| GS-9137 |
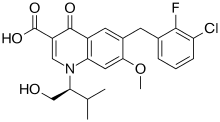
| 6-[(3-Chloro-2-fluorophenyl)methyl]-1-[(2S)-1-hydroxy-3-methylbutan-2-yl]-7-methoxy-4-oxoquinoline-3-carboxylic acid |
| (CAS 697761-98-1) |
 |
| Description: |
Elvitegravir (EVG, formerly GS-9137) is a drug used for the treatment of HIV infection. It acts as an integrase inhibitor. It was developed[1] by the pharmaceutical company Gilead Sciences, which licensed EVG from Japan Tobacco in March 2008.[2][3][4] The drug gained approval by the U.S. Food and Drug Administration on August 27, 2012 for use in adult patients starting HIV treatment for the first time as part of the fixed dose combination known as Stribild.[5] On September 24, 2014 the FDA approved Elvitegravir (tradename Vitekta) as a single pill formulation.[6]
According to the results of the phase II clinical trial, patients taking once-daily elvitegravir boosted by ritonavir had greater reductions in viral load after 24 weeks compared to individuals randomized to receive a ritonavir-boosted protease inhibitor.[7]
|
| Product No. |
KT00384 |
| Product Name |
GS-9137 |
| Synonyms |
JTK-303; EVG; D06677 |
| Formal Name |
6-[(3-Chloro-2-fluorophenyl)methyl]-1-[(2S)-1-hydroxy-3-methylbutan-2-yl]-7-methoxy-4-oxoquinoline-3-carboxylic acid |
| CAS Number |
697761-98-1 |
| Molecular Formula |
C23H23ClFNO5 |
| Formula Weight |
447.88 |
| Formulation |
A crystalline solid |
| Purity |
98%min |
| Stability |
2 years |
| Storage |
-20°C |
| Shipping |
USD45 for Europe and USA. No shipping charge once amount reach USD500 |
| Quality Control |
HNMR,CNMR,LCMS,HPLC,IR,etc. |
| Price & Availability |
In Stock. Price Negotiated. |
|
| Related Products: |
AMD 3465
Amprenavir
Atazanavir
Bevirimat
BMS-378806
BMS-538203
BMS-626529
BMS-663068
BMS-663068 Tris
BMS-707035
|
|


